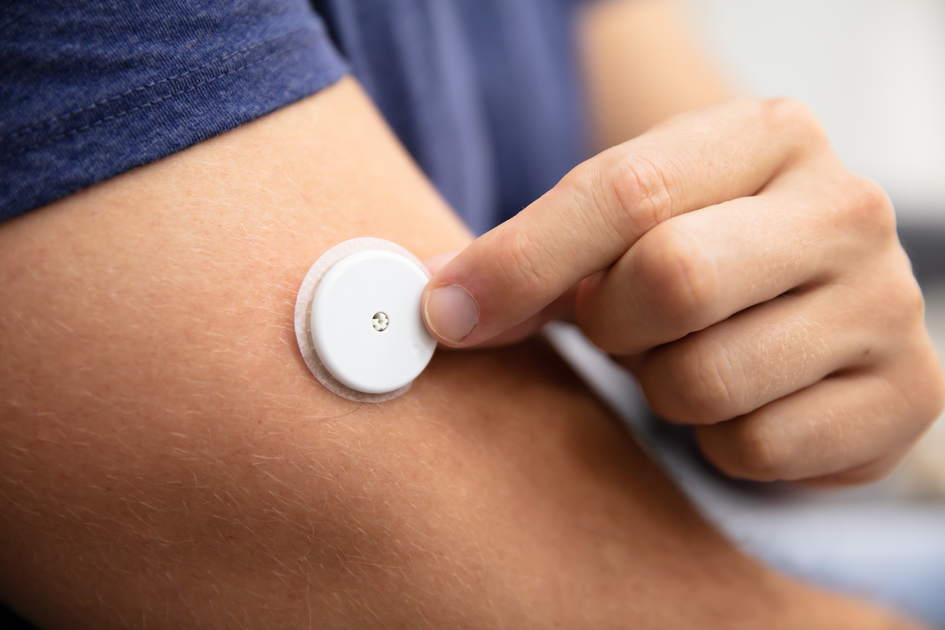
As Your Partner for What’s Next, TEAM Technologies leverages our decades of combined expertise in product development, materials, and manufacturing to continually improve time to market and, ultimately, the lives of patients and those who care for them.
TEAM Technologies is committed to Helping Those Who Help Others by providing patient monitoring devices that change patients’ lives. We develop wearable sensors for patient monitoring applications leveraging our U.S.-based manufacturing and supply chain capabilities, including ISO 13485 certification.
Patient Monitoring Solutions
Engineering and Design
Whether you have a fully engineered prototype or a new product idea sketched on a napkin, TEAM’s commercial leaders and engineers are ready to provide the creativity and expertise required to create high quality, life changing medical products. With our innovative approach to project management we can help our partners be first to market.

Prototyping
Unlike many other manufacturers, we have a fully equipped Solutions Lab dedicated to rapid prototyping and processes in place to plan for long-term DFM. Explore product designs, test alternative materials, and refine production techniques alongside our experts to help bring your products to market cost efficiently and quickly.

Converting
As an industry leader in intricate, multi-part lamination and die cutting operations, we offer a wide range of unique capabilities for the precision cutting of specialty materials, including medical adhesive films and fabrics, and other specialty membranes, plastics and materials.
- Laser Cutting on Rotary Web
- Rotary Die Cutting
- Precision Slitting
- Multi-Layer Laminations
- Flexographic/Functional Printing

Injection Molding
From custom molds for prototypes to large-scale finished products, our state-of-the-art equipment allows us to fabricate custom plastic parts that meet the precise tolerances needed for patient monitoring devices, whether you’re looking for a standalone product or need to combine a molded part with another process.

Materials Knowledge
Our deep industry knowledge of advanced materials and strong relationships with leading suppliers means we can provide the best solutions for each customer’s unique needs. Our expertise covers the full range of applications including: hypoallergenic, low-trauma, repeat insult, length of wear, weight supporting capacity, and sterilization compatibility.

Packaging and Fulfillment
As an end-to-end strategic partner, TEAM Technologies delivers on customers’ needs for high-speed, custom packaging and fulfillment. Our inventory-management services can save you time and money and simplify your supply chain so you know your product is reaching the right people at the right time.

Contact Us
About TEAM Technologies
TEAM Technologies is a leading end-to-end manufacturing solutions provider for medical and dental products. From conceptualization to completion, TEAM Technologies offers the technology and solutions for every stage of the development and manufacturing processes all under ISO 13485 and FDA regulated guidelines. TEAM Technologies’ decades of experience mean our partners can be confident products will exceed the industry’s highest quality and specification standards while being delivered on time and within budget.